The Design, Control, Monitoring and Validation of Water Systems for Pharmaceuticals, Biologics, Medical Devices, Cosmetics, and Personal Care Products

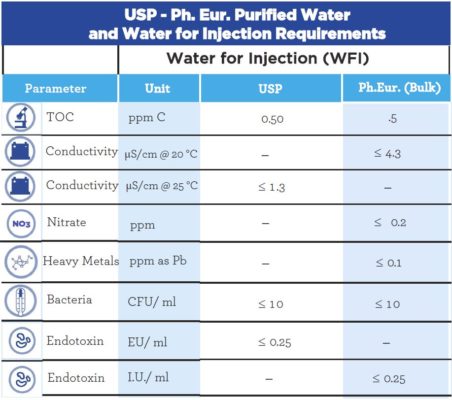
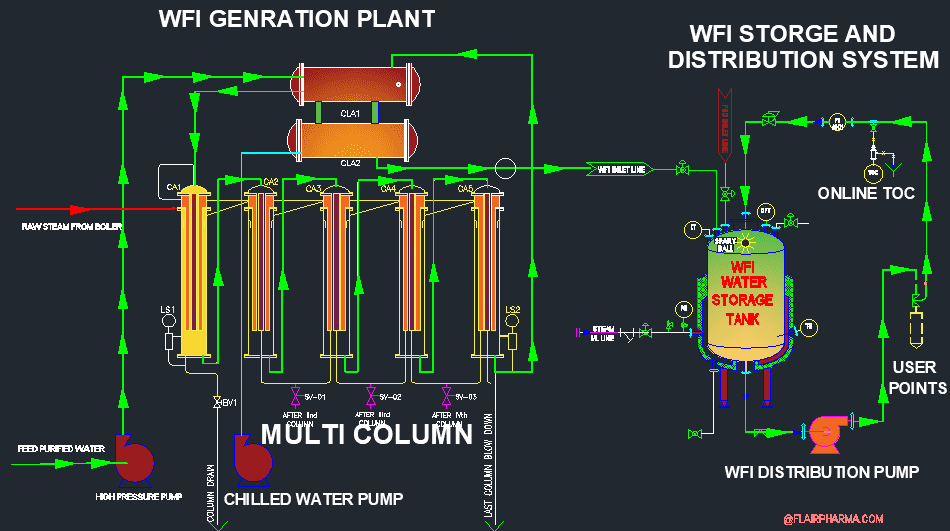
Comparison of WFI production by membrane based method & distillation based method, according to the Revised EP Monograph for WFI Production - A3P - Pharmaceutical & Biotechnology Industry

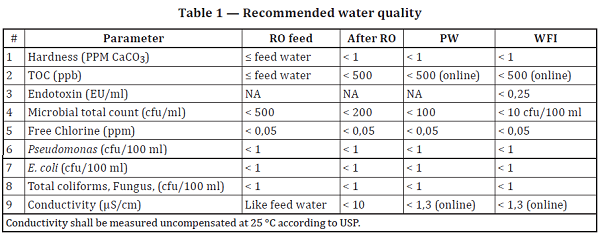
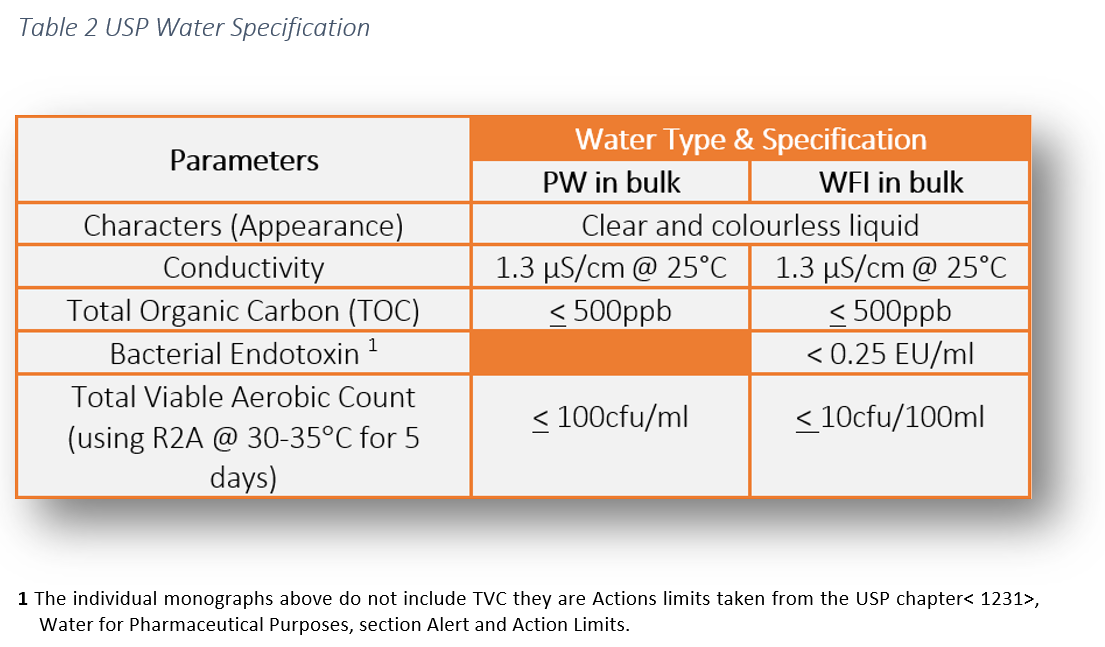
Quality specifications for different grades of water, as defined by Ph.... | Download Scientific Diagram
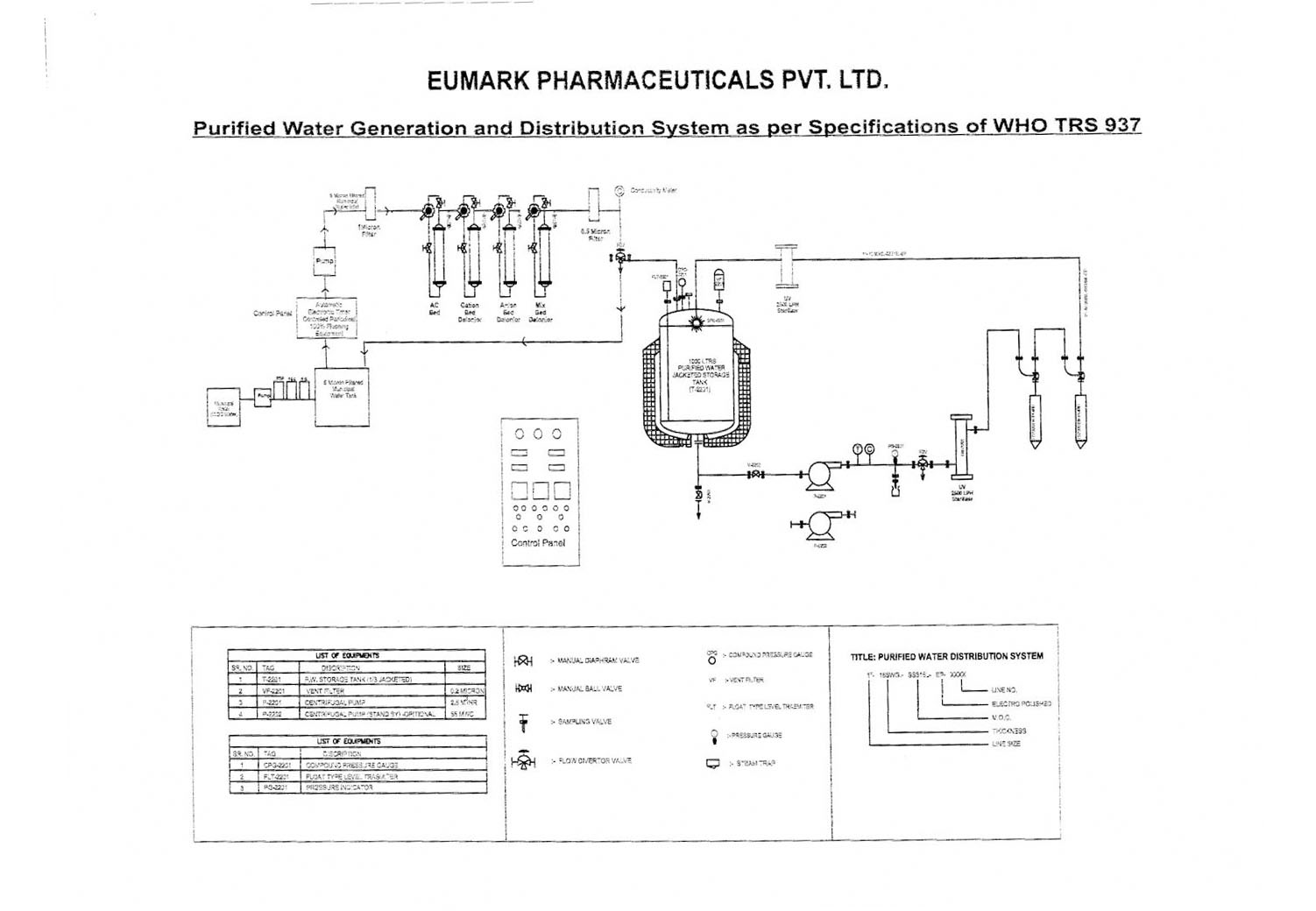
CHEMICAL QUALITY CONTROL ON WATER PRODUCED FROM WFI SYSTEM AT MEDICAL TECHNOLOGY DIVISION, NUCLEAR MALAYSIA. Muhammad Hanaffi Mo