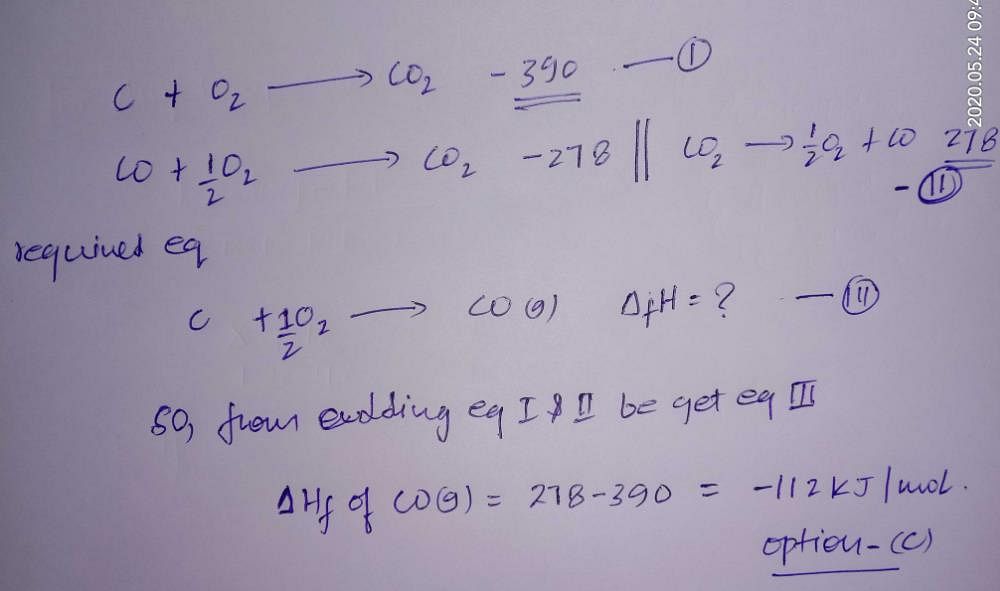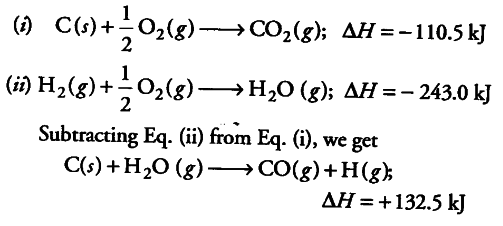66 Enthalpies of formation of co(g),CO2(g),N2O(g)and N2O4(g) are 110, 398, 81, 97 KJ/lol respectively. Find value of H for the reaction. N2O4(g) +3CO(g) — >N20(g)+3CO2(g)
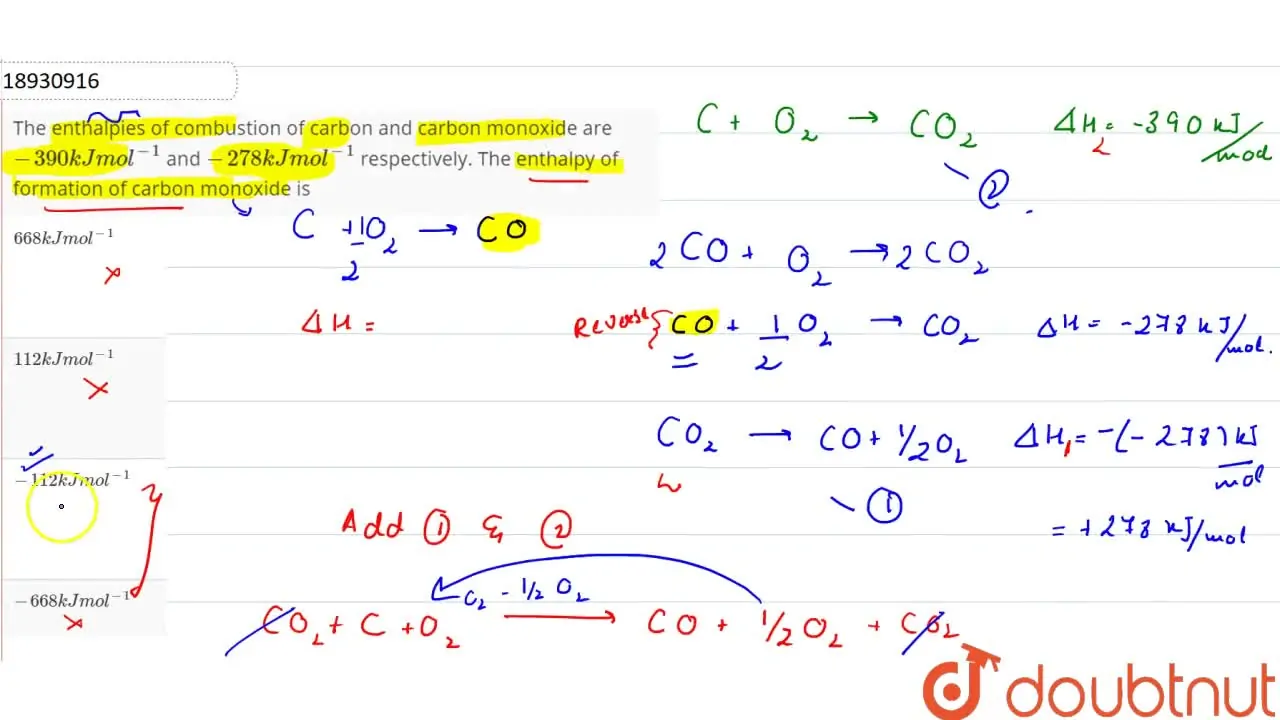
62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ

Why does the enthalpy of combustion for alkanes increase, e.g. from methane to propane, etc.? - Quora

JUHTUVUI Volume (V) Calculate the enthalpy of formation of Benzene (C Hz), standard enthalpies of combustion of Carbon (C), Hydrogen (Hz) and benzene (CH) are -393.5 Kj mol-1 - 285.83 Kj mol-

Enthalpy of combustion of carbon to C${{O}_{2}}$ is -393.5 kj/mol. Calculate heat released upon formation of 35.2g of C${{O}_{2}}$ from carbon and ${{O}_{2}}$ gas? - CBSE Class 11 Chemistry - Learn CBSE

Enthalpy of combustion of alcohols data trend graph explaining trend pattern determining delta H combustion comparison with ethers equations advanced A level organic chemistry revision notes doc brown
the enthalpies of combustion of carbon and carbon monooxide are 393.5kJ and 283kJ ,respectively the enthalpy of formation of carbon monoxide 1. 676.5kJ 2. 110.5kJ 3.110.5kJ 4.676.5

The heat of the combustion of graphite and carbon monoxide respectively are 393.5 kJ mol1 and 283 kJ mol ^{ -1 }. Thus heat of formation of carbon monoxide in kJ mol ^{ -1 } is:-110.5+110.5-55.25None of these